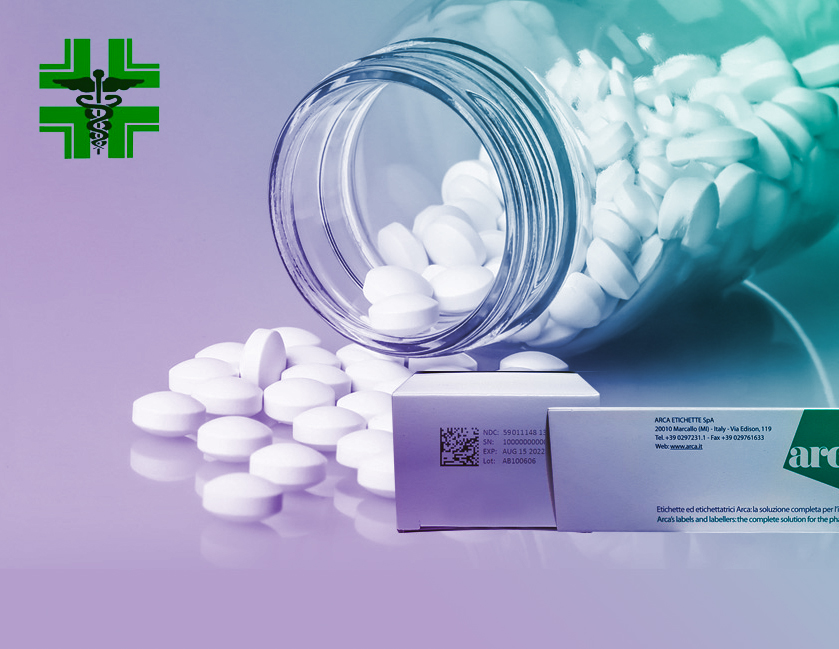
La contraffazione dei prodotti farmaceutici genera enormi rischi per la salute dei consumatori e delle grosse perdite economiche per le industrie farmaceutiche.
SERIALIZZAZIONE FARMACI
La sicurezza assoluta dei prodotti è un’esigenza per il consumatore finale e presuppone un controllo assoluto del flusso dei prodotti, dalla fabbricazione fino alla distribuzione.
È in questo contesto che molti Paesi hanno deciso di adottare delle normative volte ad introdurre elementi di sicurezza per il confezionamento di prodotti medicinali ad uso umano. Si tratta di un cambiamento significativo che garantirà l’autenticità dei farmaci e la sicurezza per i pazienti ma anche per l’industria farmaceutica e rafforzerà la sicurezza della catena di approvvigionamento dei medicinali, dai produttori alle farmacie e ospedali fino ai pazienti.
Uno dei componenti dell’anticontraffazione è sicuramente il processo di identificazione delle singole confezioni, che passa sotto il concetto di serializzazione. Ogni confezione soggetta all’obbligo di integrare le caratteristiche di sicurezza, sarà codificata con un codice 2D (GS1 Data Matrix) contenente un identificativo univoco. I dati dovranno essere stampati anche in formato leggibile dall’uomo e se è possibile adiacenti al codice a barre bidimensionale che contiene l’identificativo univoco.
In aggiunta all’identificativo univoco, l’integrità del sistema di prevenzione delle manomissioni (vedere punto 5) garantisce che la confezione non sia stata aperta o alterata dopo il rilascio dalle officine di produzione, assicurando in tal modo l’integrità del farmaco contenuto.
Attualmente, le esigenze in materia di serializzazione si limitano alla marcatura del codice univoco sul singolo prodotto venduto. Tuttavia, da qui al 2023, ogni prodotto dovrà essere tracciabile sull’insieme della catena logistica: imballaggio individuale, scatola/pallet, punto di distribuzione finale. Questo livello di serializzazione prende il nome di aggregazione.
BOLLINO FARMACEUTICO: CODIFICA, SIGILLATURA E SOFTWARE TRACCIABILITA’
I sistemi di Arca garantiscono sicurezza ed alte qualità di etichettatura. Abbiamo ideato e progettato i migliori macchinari in ambito di serializzazione farmaceutica per garantire un risultato completo.

Pharmatrack Seal 4.0

Cardboard

Sigillo antieffrazione
Normative internazionali
Unione Europea
Nel 2011 l’Unione Europea ha emesso una specifica relativa ai Medicinali Falsificati (DIRETTIVA 2011/62/EU ) che indicava i punti chiavedell’anticontraffazione che si possono semplificare nei concetti relativi alle “Safety Features” quali: Autenticità, Identificazione, Antimanomissione.a
Le regole definite negli Atti Delegati (UE) 2016-161 si applicano a decorrere dal 9 febbraio 2019 per tutti gli Stati membri dell’Unione europea con la sola eccezione di Belgio, Grecia e Italia che al momento dell’entrata in vigore della Direttiva 2011/62/EU disponevano già di sistemi per la verifica dell’autenticità dei medicinali ed identificazione delle singole confezioni (bollino farmaceutico, vedere punto 6). Per questi paesi gli articoli espressi negli Atti Delegati dovranno essere applicati al più tardi a decorrere dal 9 febbraio 2025.
La continua crescita dell’export nell’industria farmaceutica italiana, testimonia la capacità di questo settore di competere sui mercati esteri. Per continuare a crescere sarà quindi necessario procedere rapidamente verso l’adeguamento degli impianti produttivi alla Direttiva 2011/62/EU.
Resto del mondo
La serializzazione è già richiesta dai regolamenti di Paesi diversi tra loro come Argentina, Turchia (considerata il pioniere a questo proposito, che già nel 2010 ha prescritto il monitoraggio dell’intera catena di fornitura), Corea del Sud e Cina. La serializzazione farmaceutica è già realtà anche in Arabia Saudita ed Iran, dove il Data Matrix è obbligatorio dal marzo 2017 e il processo di aggregazione potrebbe essere presto integrato.
La serializzazione è obbligatoria negli USA dal novembre 2017. Ciò nonostante, la FDA ha specificato che “non verranno prese azioni nei confronti dei produttori che non si adeguano alla normativa fino a novembre 2018”. L’aggregazione sarà necessaria a partire dal 2019 e obbligatoria entro il 2023.
Le deadline più vicine
La direttiva europea sui Medicinali Falsificati (FMD) rende obbligatorio il processo di serializzazione e sigillatura Tamper Evident per ogni farmaco che entrerà nel mercato unico europeo a partire da febbraio 2019. Per questo motivo, anche alcuni paesi non membri dell’UE come Svizzera, Norvegia e Islanda hanno deciso di adeguarsi alla FMD.
Altre scadenze imminenti per l’implementazione della serializzazione farmaceutica riguardano: l’Egitto (giugno 2018), la Russia (dicembre 2019), il Pakistan (dicembre 2019), la Giordania (gennaio 2020), e il Brasile (dicembre 2021).
È previsto che gli altri membri del Consiglio di cooperazione degli Stati del golfo Persico seguano l’esempio dell’Arabia Saudita in materia di serializzazione farmaceutica.
Stesso discorso vale per l’Ucraina, che si adeguerà alle norme esistenti in Russia e UE.
In linea generale, ogni produttore farmaceutico sarà tenuto a rispettare le normative dei mercati in cui vuole vendere i suoi prodotti. Per questo motivo, le aziende farmaceutiche che esportano i loro prodotti all’estero sono soggette a queste normative anche se la serializzazione non è ancora regolata da una normativa locale.
L’identificativo unico del prodotto
Il regolamento definisce quelle che dovranno essere le caratteristiche dell’IDENTIFICATIVO UNIVOCO che sarà marcato su tutte le confezioni.
L’identificativo sarà composto da 4/5 informazioni riportate in formato leggibile e bidimensionale, in base al paese di destinazione ed alle sue specifiche norme. Avrà un suo processo di comunicazione ad una struttura multilivello di archiviazione in modo da permetterne la verifica.
• Codice che consente di identificare la denominazione comune, la forma farmaceutica, il dosaggio, le dimensioni e il tipo di confezione del medicinale;
• Una sequenza numerica o alfanumerica di non oltre 20 caratteri, generata da un algoritmo di randomizzazione
• Un numero di rimborso nazionale o un altro numero nazionale che identifica il medicinale, se richiesto dallo Stato membro in cui il prodotto è destinato ad essere immesso sul mercato;
• Il numero del lotto;
• La data di scadenza;
Come si può realizzare
Per realizzare la marcatura del codice univoco direttamente sull’astuccio farmaceutico, ci sono due tecniche principali in grado di garantire un’ottima qualità di marcatura mantenendo un’alta produttività:
– Marcatori a getto d’inchiostro
– Marcatori laser
Attualmente, la soluzione più utilizzata dalle aziende farmaceutiche è la marcatura a getto d’inchiostro. Tuttavia, sono sempre più frequenti le aziende che decidono di affidarsi alla marcatura laser, che presenta una serie di vantaggia competitivi molto interessanti:
- PRODUTTIVITA’ INEGUAGLIATA grazie alla possibilità di lavorare continuativamente, senza nessun arresto per l’approvvigionamento del materiale di consumo.
- INDELEBILITA’ La marcatura laser è per sempre.
- ECONOMIA DI ESERCIZIO Il laser non impiega nessun materiale di consumo (es: nessun inchiostro liquido).
- ALTA QUALITA’ GRAFICA sempre costante nel tempo.
- MINIMA MANUTENZIONE grazie all’assenza di parti in movimento e componenti soggetti ad usura.
- ECOLOGIA Il laser non produce residui da smaltire.
- PULIZIA ASSOLUTA La luce non sporca.
- ALTISSIMA ASPETTATIVA DI VITA fino a 100.000 ore su linee produttive non-stop.
Che cos’è un software per la tracciabilità
Per le aziende manifatturiere che operano nel settore farmaceutico è diventato obbligatorio disporre di strumenti informatici che supportino in modo rigoroso l’intero ciclo gestionale, oltre che il processo di fabbricazione.
Le caratteristiche del settore e le norme di qualità hanno spinto le aziende farmaceutiche ad estendere le procedure informatiche relative alla tracciabilità dell’intero processo produttivo, dall’acquisto delle materie prime alla produzione, dai controlli qualitativi alla distribuzione verso i depositi esterni ed i clienti.
Per gestire questo complicato processo informatico, l’industria farmaceutica si affida a delle aziende specializzate nello sviluppo di software TRACK & TRACE.
Come riportato dagli standard internazionali ANSI ISA-95 (standard dell’International Society of Automation per lo sviluppo di una interfaccia automatizzata tra sistemi aziendali e sistemi di controllo), si possono distinguere i seguenti livelli tecnologici nei sistemi di serializzazione.
Livello 0: le linee produttive delle aziende farmaceutiche (secondo ISA-95 “It defines the actual physical processes”).
Livello 1: le attrezzature di linea per la marcatura, l’etichettatura e l’ispezione come etichettatrici, marcatori inkjet o laser, sistemi di visione e verificatori di codici, stampanti desktop (secondo ISA-95 “It defines the activities involved in sensing and manipulating the physical processes”).
Arca è un produttore di dispositivi collocabili a livello 1.
Livello 2: i software di gestione e controllo della serializzazione su ciascuna linea di confezionamento (secondo ISA-95 “It defines the activities of monitoring and controlling the physical processes”).
Rende possibile la ricezione dei numeri seriali, il loro invio al marcatore e alla videocamera, l’ispezione e l’acquisizione dei dati relativi, l’aggregazione in confezioni multiple e la “rilavorazione”, l’inizio e la fine di ogni lotto produttivo e la gestione della sicurezza (CFR21 part11). Una volta terminato il processo di marcatura ed ispezione del codice univoco, il livello 2 comunica i dati di serializzazione e aggregazione ai livelli superiori.
Livello 3: il software per la gestione complessiva della serializzazione in ciascun sito produttivo (secondo ISA-95 “It defines the activities of the work flow to produce the desired end-products”).
Nel livello 3 vengono auto-generati, o ricevuti dall’esterno, i numeri seriali per ogni unità di prodotto. È una piattaforma centralizzata (software installato su un server centrale connesso a tutte le linee di confezionamento) che evita la ridondanza dei dati, facilita i back up, consente la gestione sicura dei dati di serializzazione pur garantendo autonomia operativa alle linee produttive.
Livello 4: i software di interfaccia degli ERP delle aziende farmaceutiche verso gli altri attori della supply chain e le agenzie nazionali (secondo ISA-95 “It defines the business related activities needed to manage a manufacturing organization”).
Le normative internazionali impongono a ciascuna azienda farmaceutica di mettere a disposizione tutti i dati serializzati ai produttori autorizzati (Manufacturing Authorization Holders, Contract Manufacturing Organizations), a tutti i partner della filiera (come ad esempio farmacie e gli ospedali), ai clienti e alle autorità.
Lo standard Ansi esplicita che, soprattutto in materia di sicurezza, possono determinarsi ulteriori livelli del sistema. In effetti, nell’ambito della serializzazione viene normalmente identificato anche il seguente ulteriore livello:
Livello 5: i data base centralizzati su scala nazionale o sovranazionale, come nel caso dell’Unione Europea (secondo ISA-95 “There are other non-manufacturing business-related activities that may be in levels 1 through levels 4 or higher levels, but these are not defined in this standard; for example, security activities”).
Come garantire l’integrità dell’imballaggio
Le normative in materia di anticontraffazione non indicano una metodologia specifica per la realizzazione dell’integrità dell’imballaggio. I sistemi tecnicamente disponibili comprendono: la sigillatura mediante applicazione di colla sui lembi della confezione, l’applicazione di etichette di sicurezza “tamper evident”, l’avvolgimento dell’intera confezione in una pellicola sigillata, l’utilizzo di astucci pieghevoli appositamente progettati.
La soluzione più utilizzata dall’industria farmaceutica è senza dubbio l’applicazione di etichette autoadesive Tamper Evident. Le due tecniche più comuni per il TE sono:
- Wipe Out: etichette/sigilli anti effrazione; prodotti con materiali distruttibili che si rompono se sollevati dalla confezione o dalla superficie sulla quale sono applicati, impedendone il riposizionamento.
- Etichette Void: etichette/sigilli anti effrazione; quando sollevati, lasciano sulla confezione una stampa colorata con la scritta personalizzata, il riposizionamento lascia scritte visibili.
Queste due tipologie di etichette/sigilli possono contenere elementi aggiuntivi di sicurezza:
- microtesti latenti
- inchiostri di Wood/QFX/Affirm/UV mask
- inchiostri fotocromatici e termocromatici
- inchiostri pen reactive
- stampa Guilloche e carte filigranate
Il bollino farmaceutico
Il bollino farmaceutico è un’etichetta realizzata su carta adesiva a tre strati contenente tutte le informazioni necessarie a identificare un particolare medicinale e ogni sua singola confezione.
Lo scenario in Italia
Se la prescrizione di un medicinale è a carico del SSN, il farmacista rimuove lo strato superiore del bollino, ma sulla confezione rimane una parte che ne consente l’identificazione. Inizialmente, il bollino farmaceutico includeva il codice a barre AIC (codice identificativo riferito ai farmaci destinato all’uso sugli esseri umani, assegnato all’azienda farmaceutica produttrice insieme all’autorizzazione alla vendita) e un codice a barre contenente un numero progressivo stabilito dall’Istituto Poligrafico e Zecca dello Stato in fase di stampa. A partire dal 1° gennaio 2016 l’Istituto Poligrafico e Zecca dello Stato produce un nuovo tipo di bollino farmaceutico che include un terzo codice a barre (il codice 2D Data Matrix), la cui funzione è quella di riassumere le informazioni contenute nei primi due codici. Questi codici a barre possono essere facilmente letti e acquisiti tramite una scansione con l’apposito lettore ottico. Il decreto del Ministero della Salute che regola l’utilizzo del bollino farmaceutico stabilisce inoltre le modalità di annullamento dello stesso nei casi in cui le confezioni di medicinali siano destinate a ospedali, utilizzate come campioni gratuiti, oppure per l’esportazione o lo smaltimento.
Una doppia funzione: tracciabilità e raccolta di informazioni
Il bollino farmaceutico, scansionato durante il passaggio della confezione di farmaco (a partire dal produttore per arrivare sino al paziente), viene ad ogni passo comunicato ad un’apposita Banca Dati centrale, permettendo così di individuarne l’esatta posizione in ogni momento. Oltre a garantire l’autenticità dei farmaci, questo meccanismo consente al Ministero della Salute di verificare il tipo e il numero di farmaci prescritti sulterritorio, ricavando altre informazioni utili come ad esempio il consumo di uno specifico medicinale. Le informazioni raccolte possono quindi essere utilizzate per avere una fotografia sullo stato di salute dell’intera nazione, ricavando informazioni preziose relative ad esempio alla presenza di particolari patologie, in quali aree geografiche sono maggiormente presenti, quali sono l’età e il sesso dei pazienti affetti e quale tipo di cura è stato prescritto.